 
5 (65), 10 (2017).Post-print standardized by MSL Academic Endeavors, the imprint of the Michael Schwartz Library at Cleveland State University, 2017. 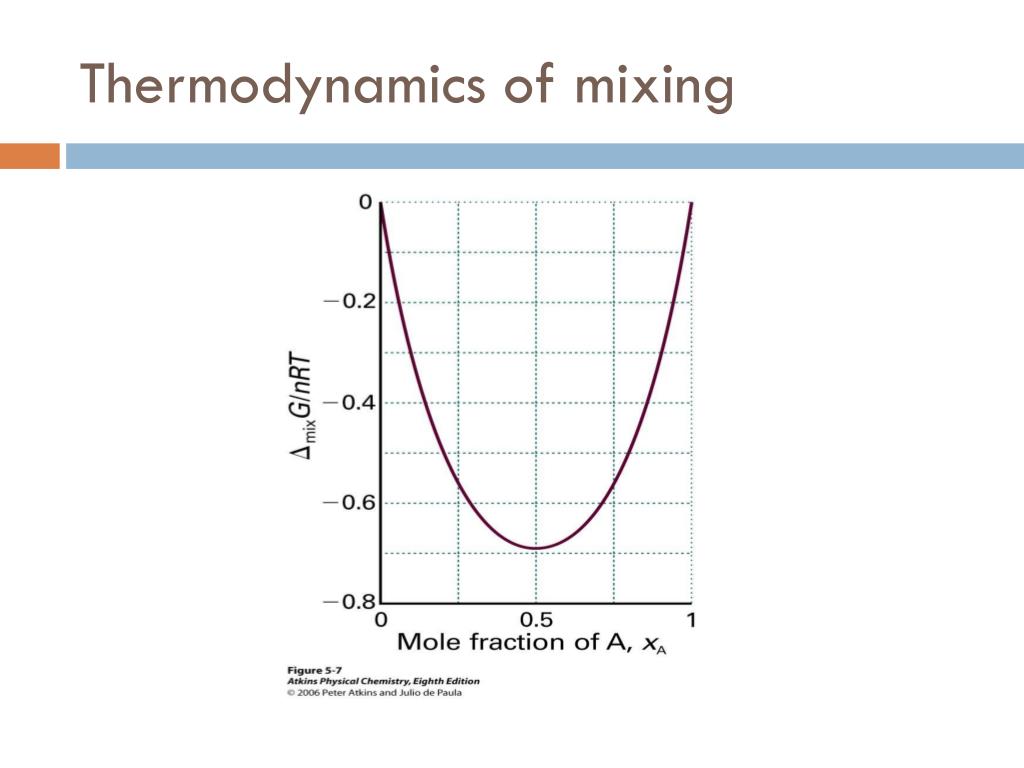
66 Fultz, B.: Vibrational thermodynamics of materials. B 52, 3280 (1995).CrossRef Google Scholar PubMed. Phonon modes and vibrational entropy of mixing in FeCr. Glukharev, “Intensification of heat transfer in the apparatus of refrigeration and cryogenic installations,” Inzh. Computational modeling of high-entropy alloys: Structures, thermodynamics and elasticity. 26, the authors summarized the configurational entropy of mixing of three equi-atomic alloys (CoCrFeNi, MoNbTaW and CoOsReRu) obtained through Monte-Carlo Molecular Dynamics (MCMD) simulations. The phase selection between solid solutions and the amorphous phase upon alloying in high-entropy alloys is intriguing. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart. 2 N2kBT V2 (2.6.3) With the partition in place, the total entropy is initially, Sinit kBln 1(E1 V1 N1)2(E2 V2 N1) S1(E1 V1 N1) +S2(E2 V2 N2) (2.6. In the earlier posted question named Derive expression for internal energy of mixing and entropy of mixing using statistical thermodynamics the entropy of mixing was found by assuming an ideal solution. In the recent review article of Gao et al. Meanwhile, the high mixing entropy also favors the amorphous phase formation. This is qualitatively easily visualised in terms. Ram Nandan Singh 1, Victor Witusiewicz Add to Mendeley Cite (01)01272-5 Get rights and content Abstract The excess entropy of formation of binary alloys has been described as the sum of configurational, vibrational, electronic and magnetic contributions. Introduction to Chemical Engineering Thermodynamics. When two pure substances mix under normal conditions there is usually an increase in the entropy of the system. In Thermodynamics and an Introduction to Thermostatistics 2nd Edition, Callen states that the Entropy of a mixture of ideal gases is given by (3.39) S j N j s j 0 + ( j N j c j) R l n T T 0 + j N j R l n ( V N j v 0) He then states that we can re-write it as follows (3. These are the relative volumes of the molecules. In addition, we can see that /1 T and thus the energy of mixing will vary with temperature. Three factors which may contribute to the excess entropy of mixing of binary solutions have been explored. Karakulov, “Analysis of losses in the evaporative-condenser circuit of a central air conditioner using the entropy-statistical method,” Kholod. What is the entropy of mixing of 1 mol of O2 with 1 mole of N2 at 25oC, assuming that they are ideal gases final answer: 11.53 J K-1. Furthermore, we can see that the favorable entropy of mixing decreases as the degree of polymerization of either or both species increases that is polymers are less likely to mix if either species is very large. Shishov, “Analysis of losses in the heat exchanger – heat reclaim unit using the entropy-statistical method,” Kholod. Lavrov, “Analysis of losses in ventilation installations using the entropy-statistical method,” Kholod. Talyzin, “Entropy-statistical analysis of low-temperature refrigeration cycles and the choice on its basis of the optimal refrigeration system for a store,” Kholod. Talyzin, “Practical application of the entropy-statistical method for the analysis of refrigeration cycles,” Kholod. gases, in whose equation of state no members occur which depend on. Shishov, “Entropy-statistical analysis of classical refrigeration cycles for air conditioning systems,” Kholod. If we now calculate the change of entropy which occurs on mixing two ideal gases ( i.e. 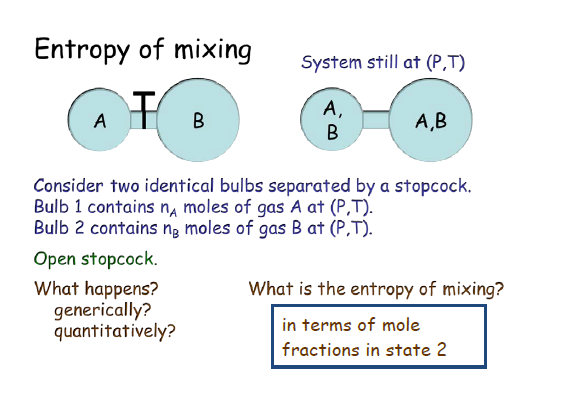
Arkharov, “A unified thermodynamic temperature space, heat, cold, exergy, and entropy as basic concepts of engineering cryology,” Kholod. Sychev, “Again about entropy and about the problem of determining the real (actual) values of energy losses due to irreversibility,” Kholod. Lavrov, “Results of calculations of energy losses in cooling processes using entropy-statistical analysis using temperature averaging and taking into account its change,” Vest. Lavrov, “Entropy production and exergy loss during mixing of gases,” Khimich. Liquid-liquid phase separation is a phenomenon commonly observed in vinaigrette salad dressing, which is made from a mixture of oil and vinegar. Entropy-Statistical Analysis of Low-Temperature Systems, Izd-vo MGTU im. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
